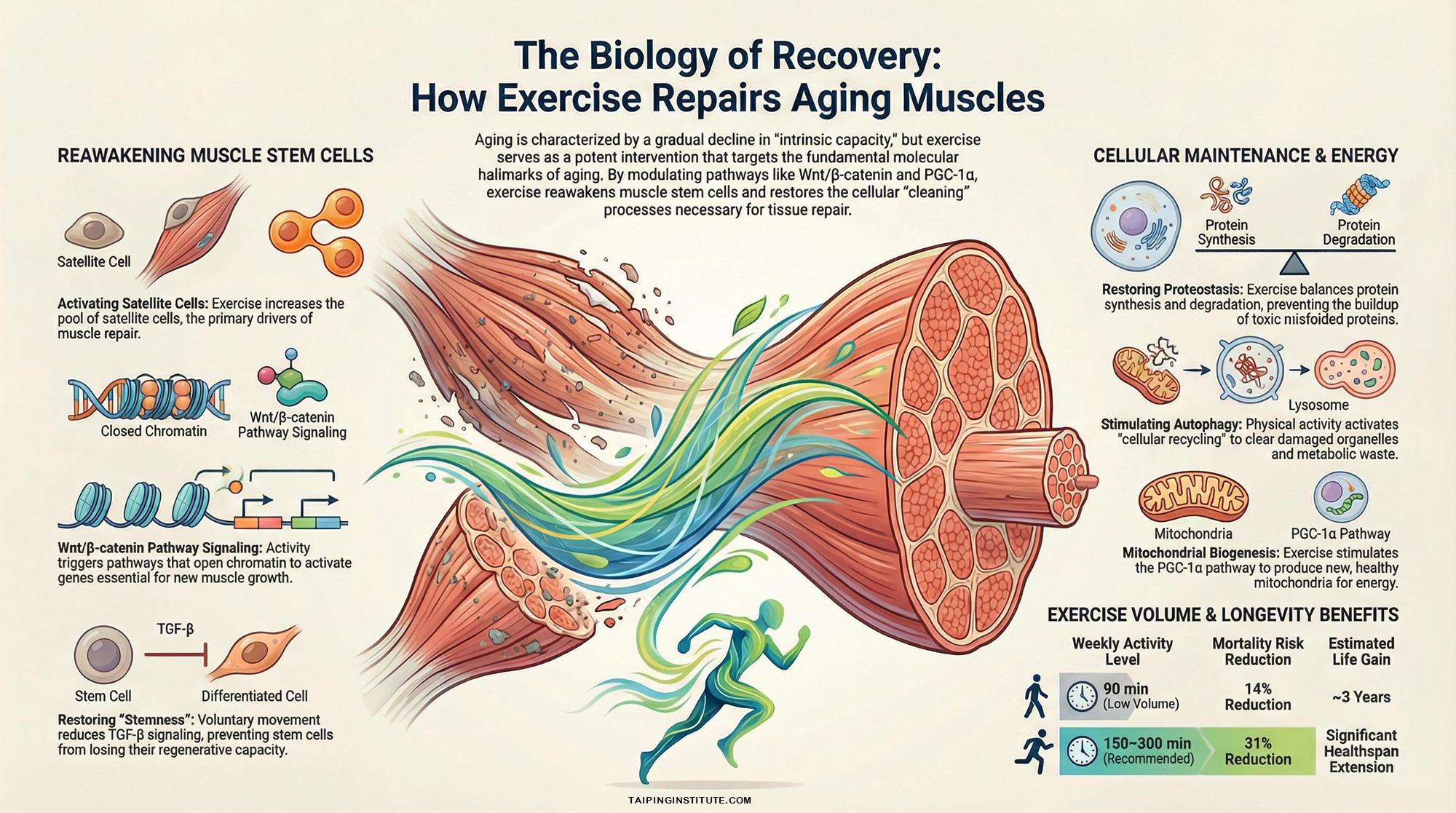
One of the most visible signs of aging is the loss of muscle mass and regenerative potential, a process driven by stem cell exhaustion. In aging muscles, the pool of satellite cells—the stem cells responsible for tissue repair, gradually declines. Research shows that long-term exercise can increase this pool and stimulate the Wnt/β-catenin signaling pathway, which is essential for activating these cells to repair damaged fibres. Furthermore, exercise reduces the burden of cellular senescence, a state where “zombie” cells stop dividing but remain in the body, secreting inflammatory factors that damage surrounding healthy tissue. By suppressing these senescence programs, exercise allows muscle environments to remain more youthful and resilient.
At a deeper molecular level, exercise serves as a rigorous “housekeeping” service for our cells through the maintenance of proteostasis and the stimulation of autophagy. With age, the body’s ability to recycle damaged proteins and organelles falters, leading to toxic cellular waste. Exercise activates pathways like AMPK and suppresses harmful molecules such as miR-34a, which otherwise impair these natural cleaning processes. By restoring autophagic flux, exercise ensures that muscle cells can effectively clear away debris and misfolded proteins that would otherwise lead to tissue dysfunction and disease.
Finally, exercise revitalises the cell’s power plants, the mitochondria, which typically decline in efficiency as we age. Regular activity promotes mitochondrial biogenesis and restores respiratory function while reducing the production of harmful reactive oxygen species (ROS). This is often accompanied by epigenetic rejuvenation, where exercise-induced modifications to DNA methylation and histone structure “reset” gene expression to a more youthful signature. Even modest amounts of activity, such as 15 minutes of daily exercise, have been shown to provide significant longevity benefits, proving that it is rarely too late to start repairing the damage of time.
As the global population of individuals over 60 years old continues to grow, a major public health challenge has emerged: the gap between living longer and staying healthy. While medical advances have extended lifespans, they have not always preserved “healthspan”, the years spent free from chronic disease and functional decline. Regular physical exercise has emerged as a critical, cost-effective strategy to bridge this gap, acting not merely as a fitness tool but as a powerful biological intervention that can partially reverse the molecular “hallmarks” of aging. By targeting the fundamental mechanisms of decay, exercise helps maintain the intrinsic capacity of our bodies to move and function.
One of the most visible signs of aging is the loss of muscle mass and regenerative potential, a process driven by stem cell exhaustion. In aging muscles, the pool of satellite cells, the stem cells responsible for tissue repair, gradually declines. Research shows that long-term exercise can increase this pool and stimulate the Wnt/β-catenin signaling pathway, which is essential for activating these cells to repair damaged fibres. Furthermore, exercise reduces the burden of cellular senescence, a state where “zombie” cells stop dividing but remain in the body, secreting inflammatory factors that damage surrounding healthy tissue. By suppressing these senescence programs, exercise allows muscle environments to remain more youthful and resilient.
At a deeper molecular level, exercise serves as a rigorous “housekeeping” service for our cells through the maintenance of proteostasis and the stimulation of autophagy. With age, the body’s ability to recycle damaged proteins and organelles falters, leading to toxic cellular waste. Exercise activates pathways like AMPK and suppresses harmful molecules such as miR-34a, which otherwise impair these natural cleaning processes. By restoring autophagic flux, exercise ensures that muscle cells can effectively clear away debris and misfolded proteins that would otherwise lead to tissue dysfunction and disease.
Finally, exercise revitalises the cell’s power plants, the mitochondria, which typically decline in efficiency as we age. Regular activity promotes mitochondrial biogenesis and restores respiratory function while reducing the production of harmful reactive oxygen species (ROS). This is often accompanied by epigenetic rejuvenation, where exercise-induced modifications to DNA methylation and histone structure “reset” gene expression to a more youthful signature. Even modest amounts of activity, such as 15 minutes of daily exercise, have been shown to provide significant longevity benefits, proving that it is rarely too late to start repairing the damage of time.