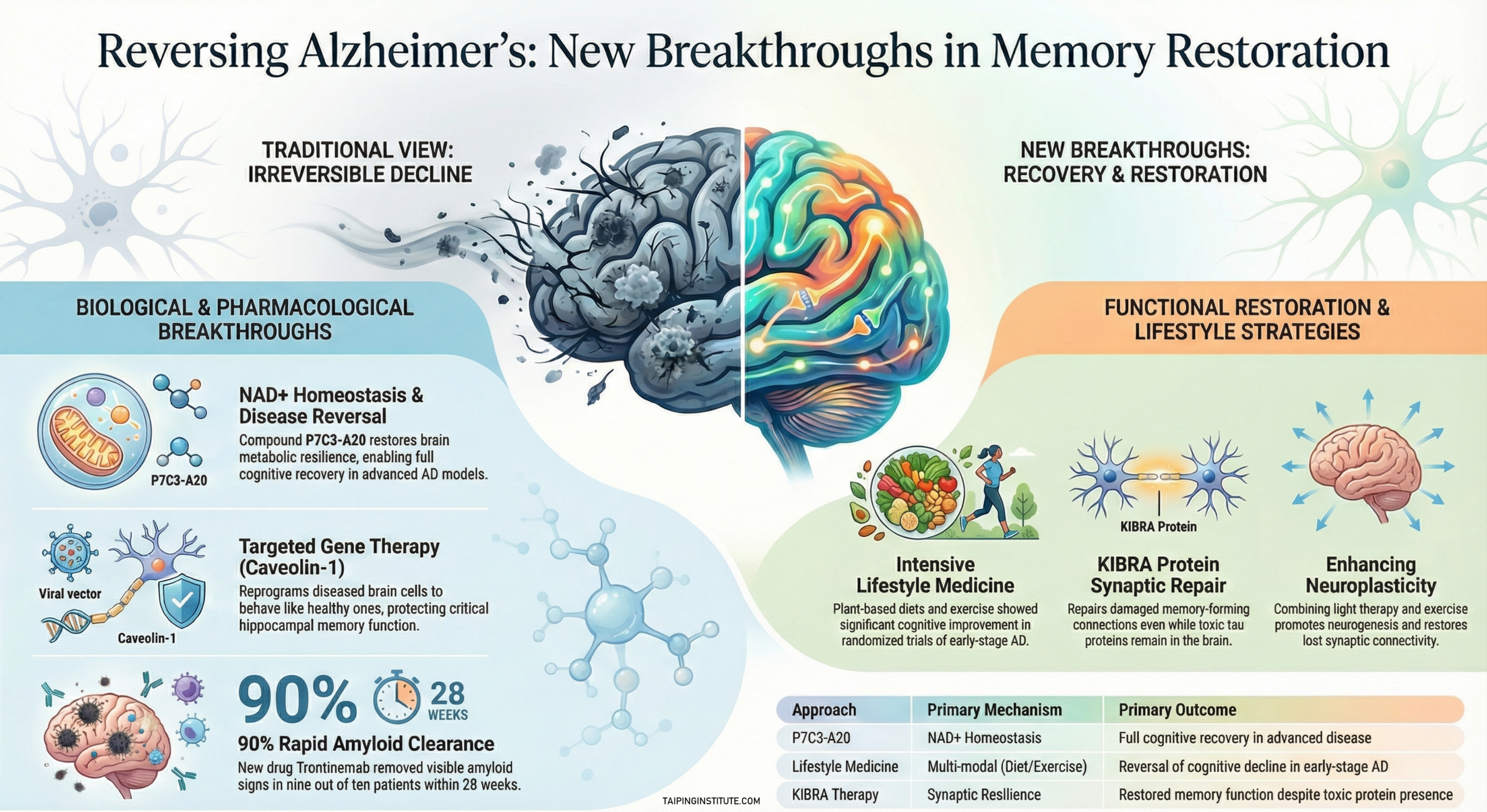
For over a century, Alzheimer’s disease has been considered a one-way street of cognitive decline, but recent scientific breakthroughs are beginning to challenge the deep-seated dogma of irreversibility. A landmark study has provided proof of principle that restoring metabolic balance in the brain can not only halt but actively reverse advanced Alzheimer’s pathology in animal models. By focusing on “brain resilience”, the organ’s intrinsic ability to adapt and recover from injury, researchers are opening a new frontier in memory restoration that moves beyond merely masking clinical symptoms.
At the heart of this shift is the discovery that disrupted brain NAD+ homeostasis is a central mechanism driving the loss of cognitive function. Researchers found that the severity of Alzheimer’s in both humans and mice correlates directly with the depletion of this vital metabolic currency. Compellingly, treating mice at advanced stages of the disease with a compound called P7C3-A20 restored NAD+ levels to normal, leading to a full recovery of cognitive function and memory. This intervention successfully repaired the blood-brain barrier, reduced neuroinflammation, and enhanced the survival of young hippocampal neurons, suggesting that even established damage can be rectified.
Complementary breakthroughs are now moving away from solely clearing toxic proteins toward directly repairing the brain’s damaged architecture. Scientists at the Buck Institute have identified a protein called KIBRA, which is essential for synapses to form memories; by administering a functional version of this protein, they were able to repair synaptic plasticity and restore memory in mice despite the continued presence of toxic tau. Similarly, experimental gene therapies are being developed to “reboot” the brain by reprogramming diseased cells to behave like healthy ones, effectively restoring function to circuits that were previously thought to be lost.
These laboratory successes are increasingly mirrored by clinical progress, with 2025 appearing to be a turning point for human treatment. Long-term data for current drugs like lecanemab and donanemab now show that memory and thinking can be stabilised for up to four years in early-stage patients, giving them precious time back with loved ones. Furthermore, next-generation treatments like trontinemab are using “brain shuttle” technology to cross the blood-brain barrier more effectively, clearing amyloid plaques with unprecedented speed and fewer side effects. The introduction of blood-based biomarker tests to the NHS by 2029 could further revolutionise this field by allowing for earlier, more accurate diagnoses.
Finally, the role of intensive lifestyle medicine is providing a crucial third pillar for memory restoration. A phase 2 clinical trial has demonstrated that a whole-food, plant-based diet combined with regular exercise and stress management can improve cognition and function in individuals already diagnosed with early-stage Alzheimer’s. As our understanding of neuroplasticity and the brain’s metabolic requirements deepens, the combined force of pharmacological, genetic, and lifestyle interventions offers the first genuine hope that the most feared condition of later life may one day be treatable, or even reversible.